Chromium – Atomic Radius – Cr Element Chromium Atomic Number 24 Symbol Cr Element Category Transition Metal Phase at STP Solid 2 more rows. The atomic radius is defined as one-half the distance between the nuclei of two identical atoms that are bonded together. If the atomic radius of Cris 1.25 Å, what is the density of Cr metal in g/cm? All values of radii are given in picometres (pm). Consider a cubic array of bromide ions in which the anions are touching along the face of the cube. As you move down a column or group, the ionic radius increases.
Potassium atomic radius pdf#
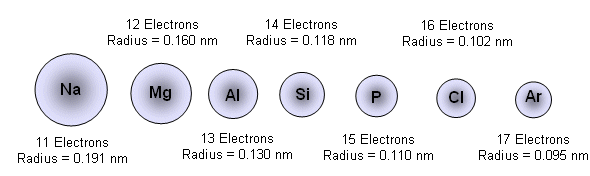
6 Why do the properties of elements change in a regular way across a period of time? Request PDF | Size effect on magnetic properties of MnCr2O4 nanoparticles | This paper studies the size effect on the magnetic properties of spinel MnCr2O4 powders synthesized by … The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Atomic radii vary in a predictable way across the periodic table. Assuming that the atomic radius of Chromium is 1.25 angstroms ( 10^-10 m) The edge length = 2.887 angstroms For a cube the volume is (edge length)^3 = 2.582*10^-23 Atom properties of chromium. Atomic radii decrease, however, as one moves from left to right, across the Periodic Table. Depending on the definition, the term may apply only to isolated atoms, or also to atoms in condensed matter, covalently bound in … Radioactive Chromium-51 is used as a tracer in the diagnosis of blood volume. We see that there is 1 atom per unit cell ( 1 8atom at each corner) and that the edge length of the cell ( a) is twice the atomic radius ( r ). Analysis of the powder XRD data indicates a cubic cell (a. Beside this, what are SI units of atomic radius? Ionic radius, r ion, is the radius of a monatomic ion in an ionic crystal structure. Atomic radii are often measured in angstroms (Å), a non-SI unit: 1 Å = 1 × 10 − 10 m = 100 pm. Therefore, there are various non-equivalent definitions of atomic radius.Stainless steel contains 12-18% chromium and usually about 8% nickel.

Potassium atomic radius free#
However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Potassium atom is 203pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Potassium are 39 41. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z.

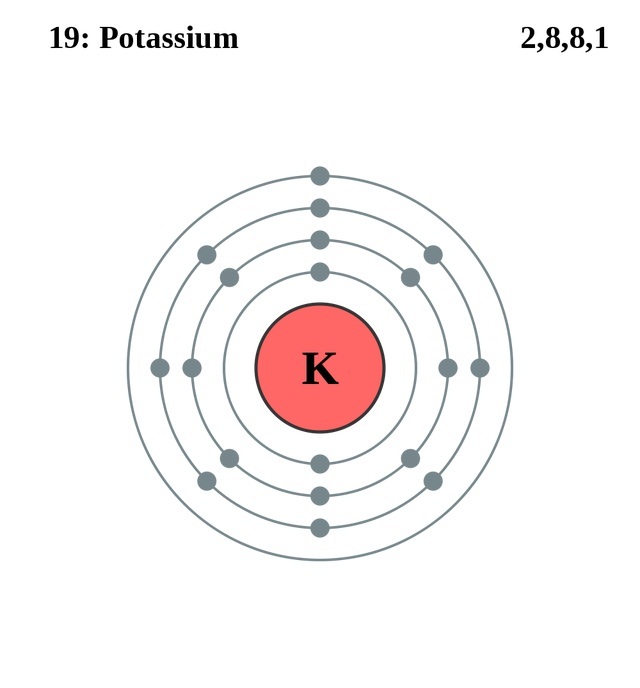
Potassium is a chemical element with atomic number 19 which means there are 19 protons in its nucleus.
Atomic Number – Protons, Electrons and Neutrons in Potassium
